Benzene Phase Diagram
With no invariant sections no front cover texts and no back cover textsa copy of the license is included in the section entitled gnu free documentation license. Permission is granted to copy distribute andor modify this document under the terms of the gnu free documentation license version 12 or any later version published by the free software foundation.
Therefore from the phase diagram this mixture is outside the area of the binodal curve.

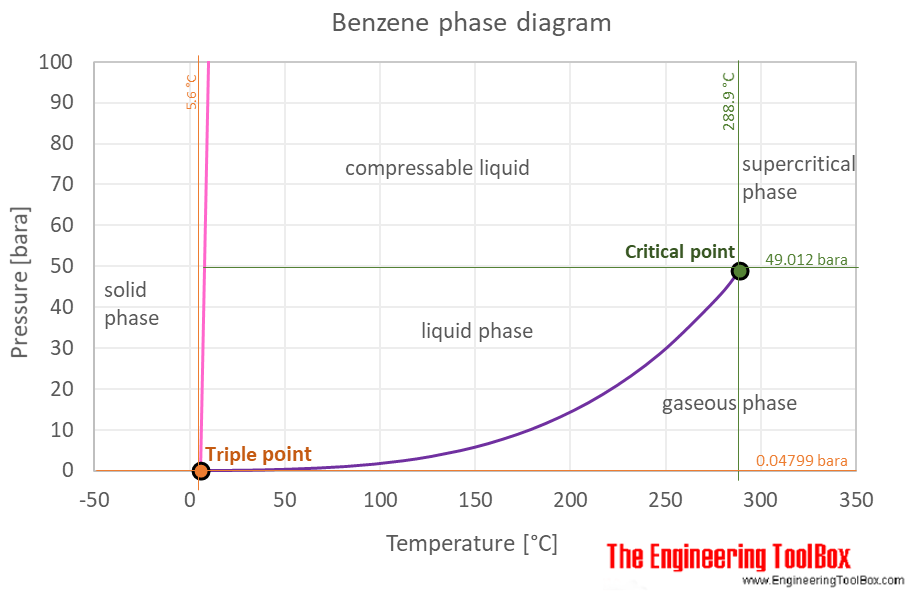
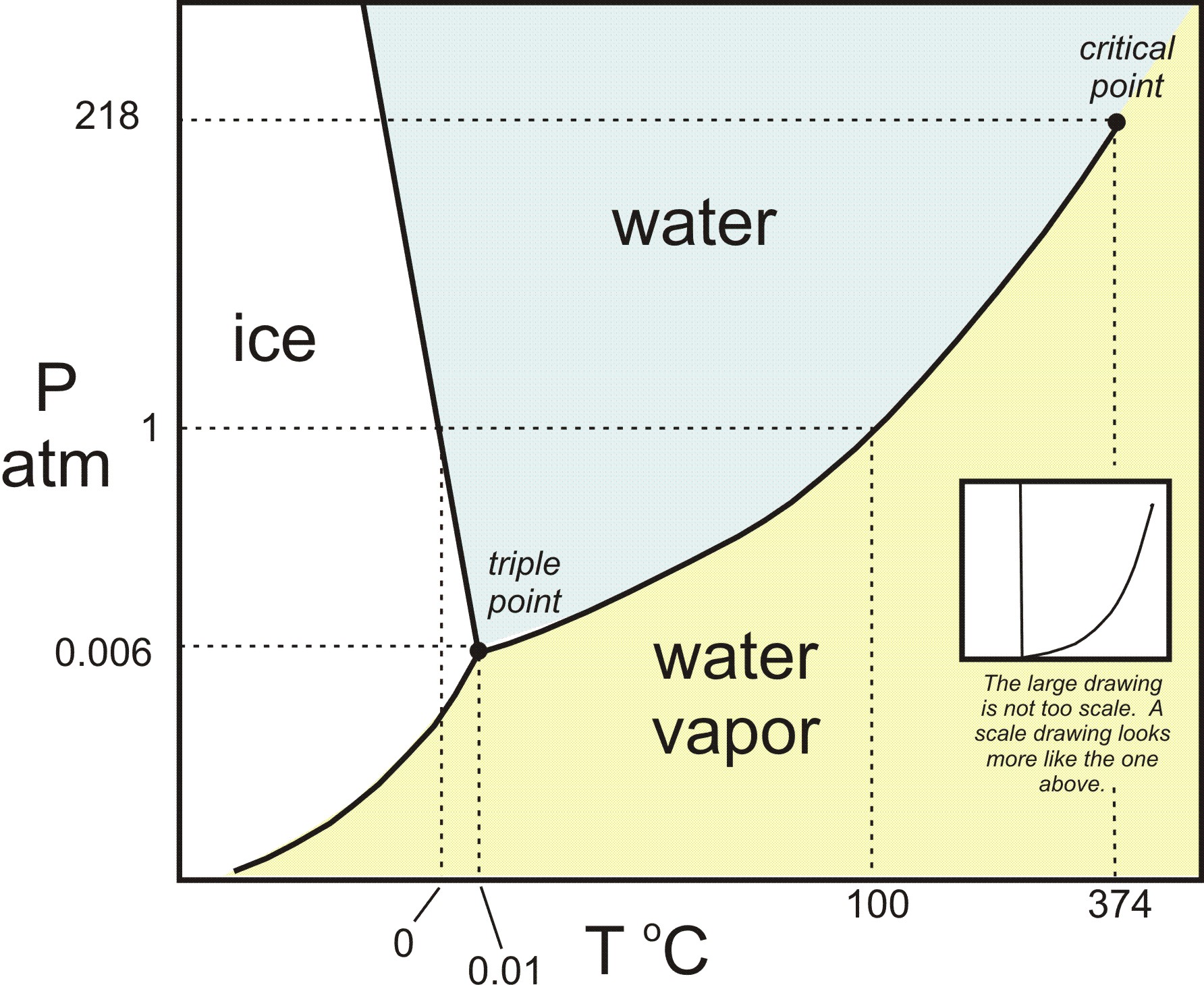
Benzene phase diagram. Vapor pressure diagrams and boiling diagrams we are now ready to begin talking about phase diagrams involving two. Phase diagram of water also exhibits a rich variety of different structures the ice polymorphs not shown in the phase diagram above. The curve between the critical point and the triple point shows the benzene boiling point with changes in pressure.
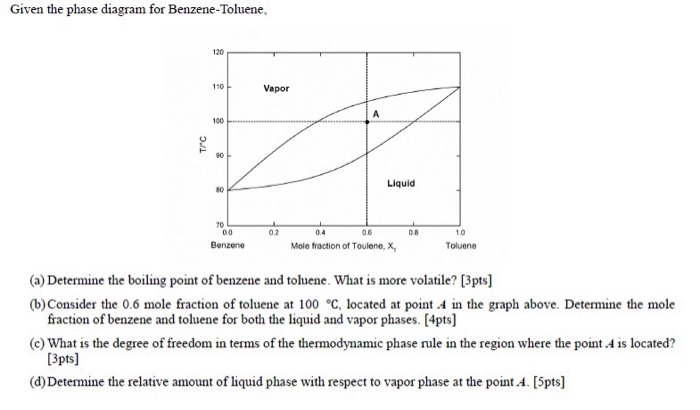
Law vapor pressure of benzene. For a binary mixture in two phases the degree of freedom f is given by f m π 2 2 2 2 2 41 1 as an example we will use the benzene toluene vapor liquid system. The solid line from tol vp to bz vp is the total vapor pressure of the solution which is just the sum of the two raoults law vapor pressures the sum of two straight lines is a straight line.
Therefore a clear single liquid phase of solution is formed. The water phase diagram is shown in the figure below. Phase diagram of water.
These triple points are at 12 05 kbar and 204 5c and 225 05 kbar and 335 5c respectively. Ethanol 475 x 100 94 water 025 x 100 4 toluene 015 x 100 2 therefore from the phase diagram this mixture is outside the area of the binodal curve. Here we see the slight slope to the left of the solid liquid coexistence line.
The phase diagram of benzene has been determined to 35 kbar. As with benzene the solid region of the fig. Two triple points liquidbenzene ibenzene ii and liquidbenzene iibenzene iii have been located.
Chapter 4 distillation 41 vapor liquid equilibrium relations the equilibrium in vapor liquid equilibrium is restricted by the phase rule. The phase diagram for benzene shows the phase behavior with changes in temperature and pressure.
 Figure 6 From Adsorption Of Benzene On The Ruo 2 110
Figure 6 From Adsorption Of Benzene On The Ruo 2 110
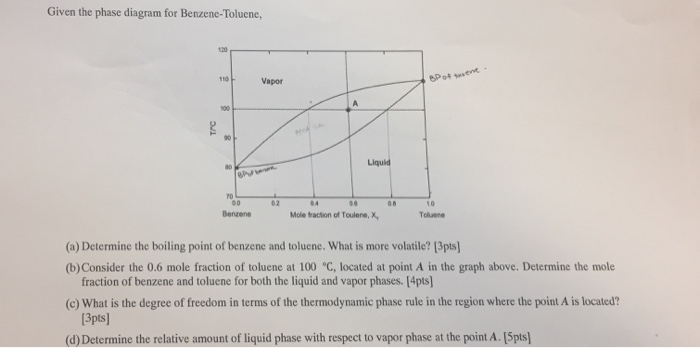
Lever Rule Applied To The Benzene Toluene Vapor Pressure
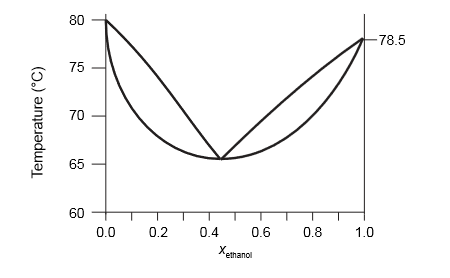 A Temperature Versus Composition Phase Diagram For
A Temperature Versus Composition Phase Diagram For
 Benzene Thermophysical Properties
Benzene Thermophysical Properties
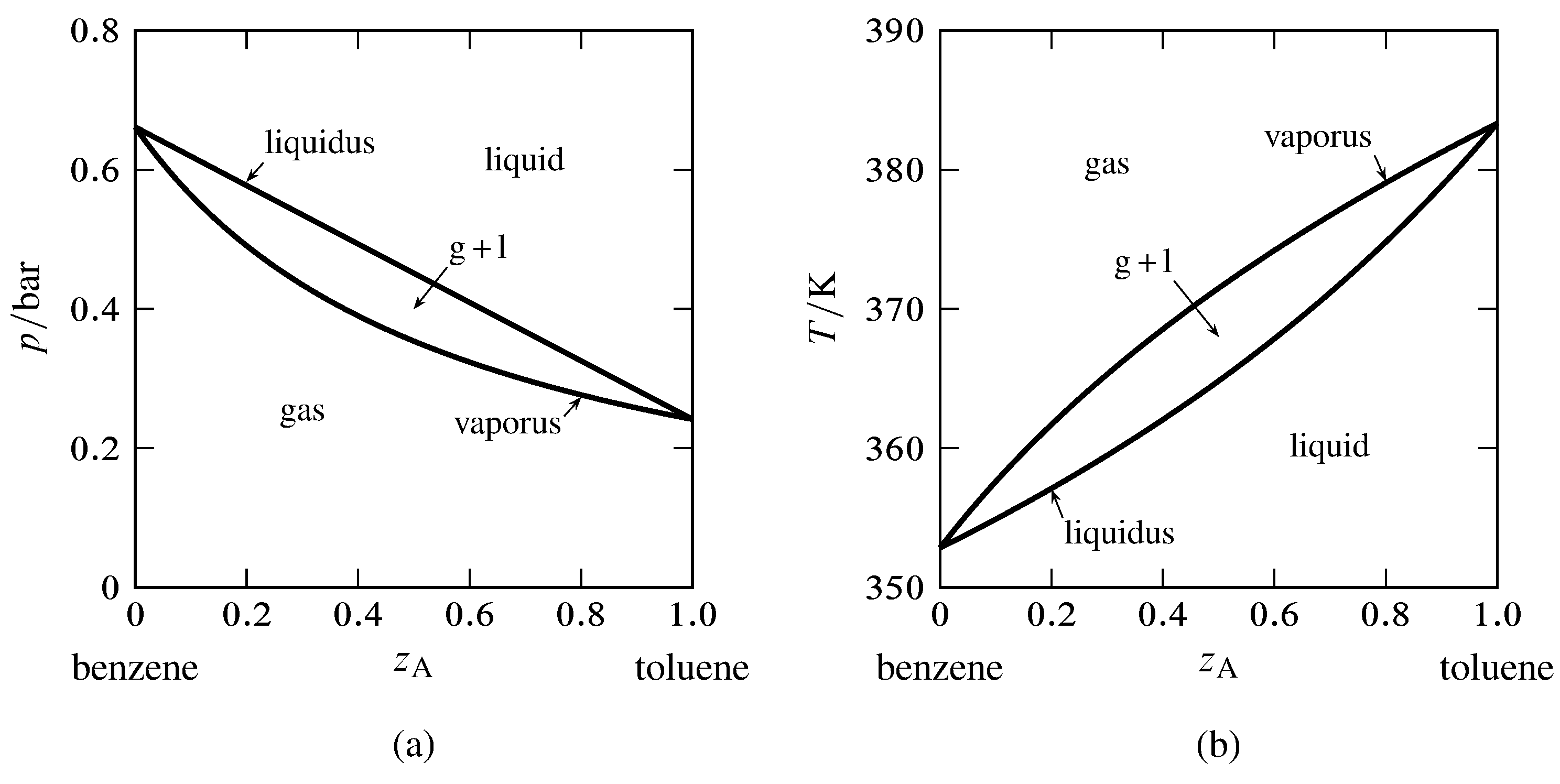 13 2 Phase Diagrams Binary Systems Chemistry Libretexts
13 2 Phase Diagrams Binary Systems Chemistry Libretexts
 Phase Diagram Of Benzene Propene At A Mole Ratio Of
Phase Diagram Of Benzene Propene At A Mole Ratio Of
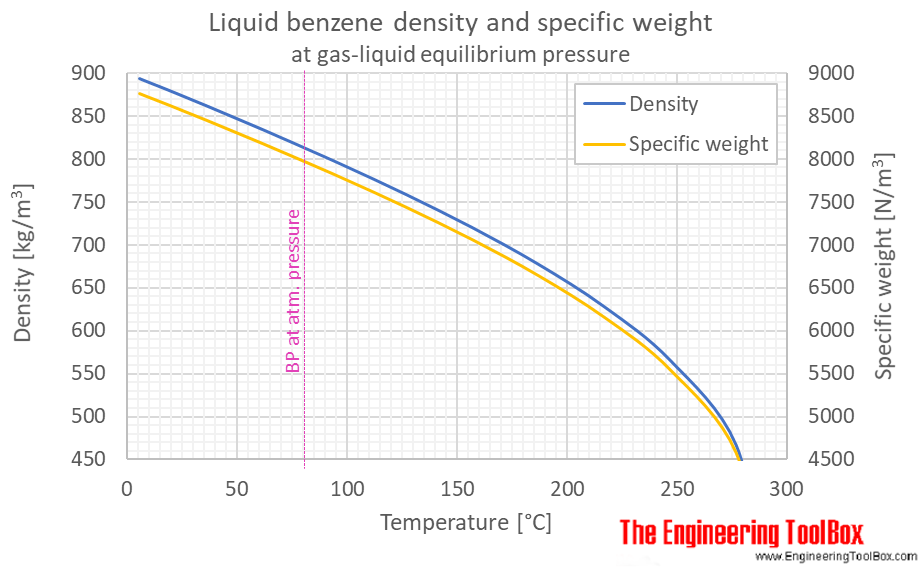 Benzene Density And Specific Weight
Benzene Density And Specific Weight
Explain The Three Liquids Ternary Phase Diagram Chemistry
Benzene Under High Pressure A Story Of Molecular Crystals
 Condensed Phase Behaviour Of Ionic Liquid Benzene Mixtures
Condensed Phase Behaviour Of Ionic Liquid Benzene Mixtures
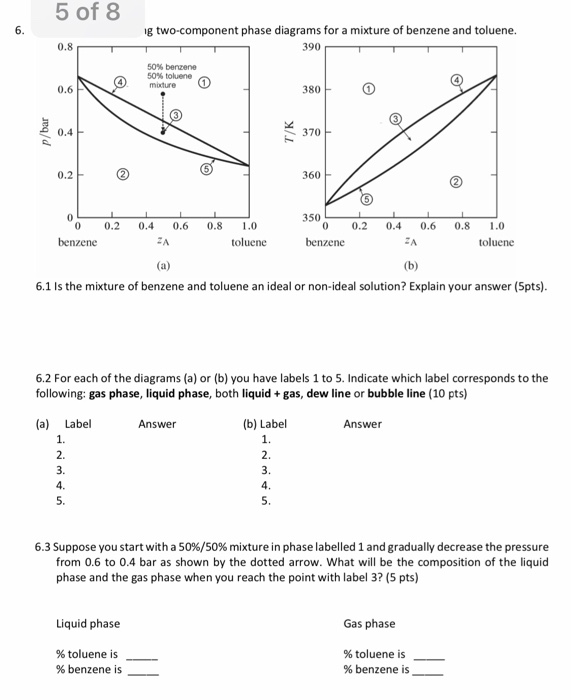 5 Of 8 G Two Component Phase Diagrams For A Mixtur
5 Of 8 G Two Component Phase Diagrams For A Mixtur
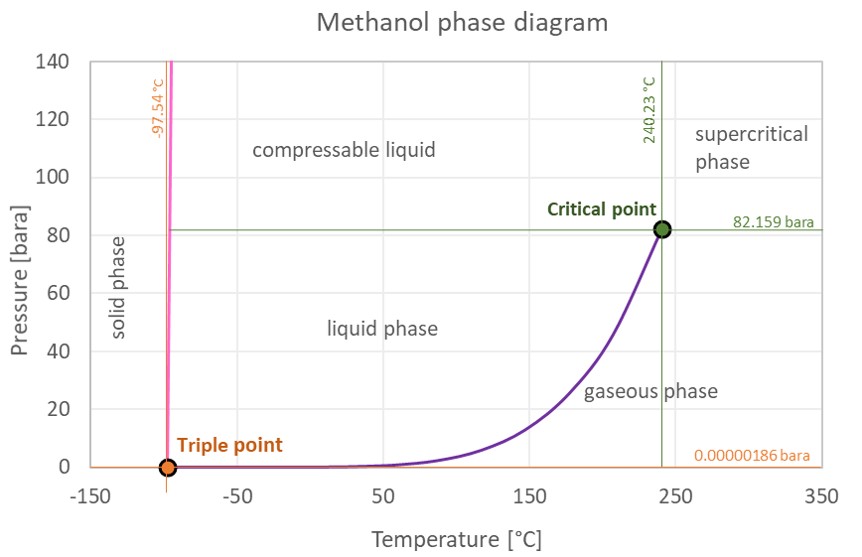 Methanol Thermophysical Properties
Methanol Thermophysical Properties
 If You Follow The Vaporization Curve Up To High Temperature
If You Follow The Vaporization Curve Up To High Temperature
Introduction To Chemical Engineering Processes Vapor Liquid
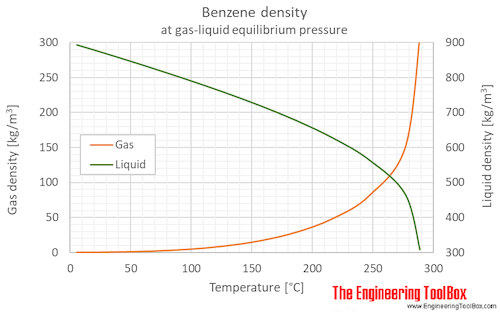 Benzene Density And Specific Weight
Benzene Density And Specific Weight
 Binary Phase Diagrams For Pentane C5h12 Benzene C6h6
Binary Phase Diagrams For Pentane C5h12 Benzene C6h6
A Ternary Phase Diagram For A Less Hazardous System
 Model To Describe The Binodal Curve On A Type 1 Ternary
Model To Describe The Binodal Curve On A Type 1 Ternary
Phase Diagrams For Three Component Mixtures In
 Experimental Phase Diagram Of Benzene 1 Download
Experimental Phase Diagram Of Benzene 1 Download



Programming assignment help
BalasHapusThetutorshelp.com For any kind of Programming Assignment Help, we at Programming Homework Helper are always ready to assist you. We believe in excellent learning and provide accuracy while solving programming assignments. To get Programming Assignment Help, https://www.thetutorshelp.com/programming-assignment-help.php
Programming assignment help
c programming assignment help
BalasHapusDo you need C Programming Help? Our C Programming Assignment Help experts are available 24/7 for your help. Get instant online C Programming Help now! https://www.thetutorshelp.com/c-programming-assignment-help.php
c programming assignment help
R Programming Assignment Help
BalasHapusDo you need R Programming Assignment Help? Our R Programming Assignment Help experts provide the best Online R Assignment Help within the given deadline. Get help now!https://www.thetutorshelp.com/r-programming-assignment-help.php
R Programming Assignment Help