Use The Mo Diagram Provided Below To Answer The Following Questions
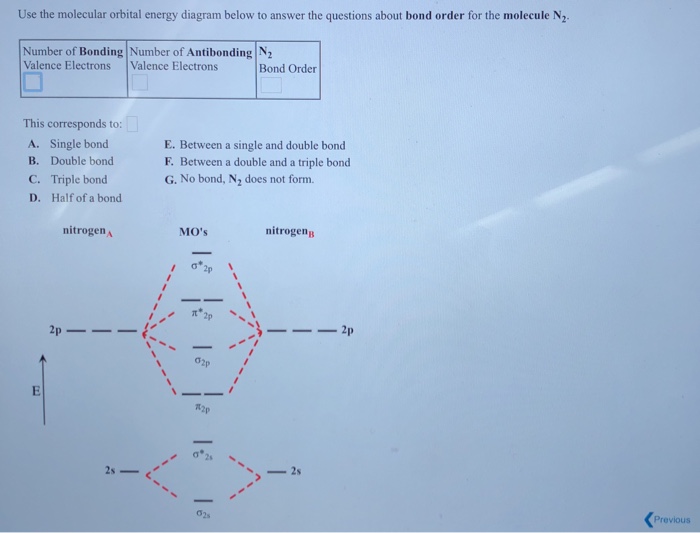
Select is n2 paramagnetic or diamagnetic select what is the bond order for not. I have attached 2 photos to help answer this question.
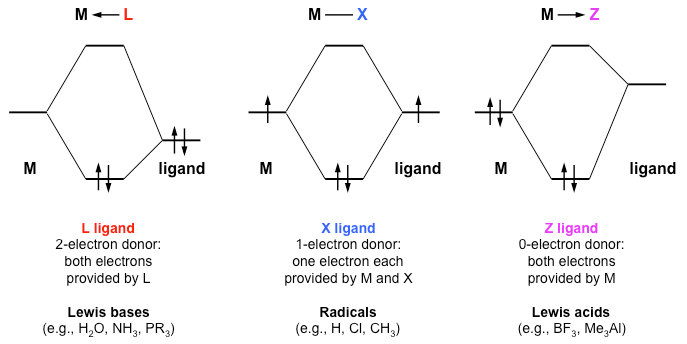 Introduction To Inorganic Chemistry Coordination Chemistry
Introduction To Inorganic Chemistry Coordination Chemistry
Gas liquid solid.
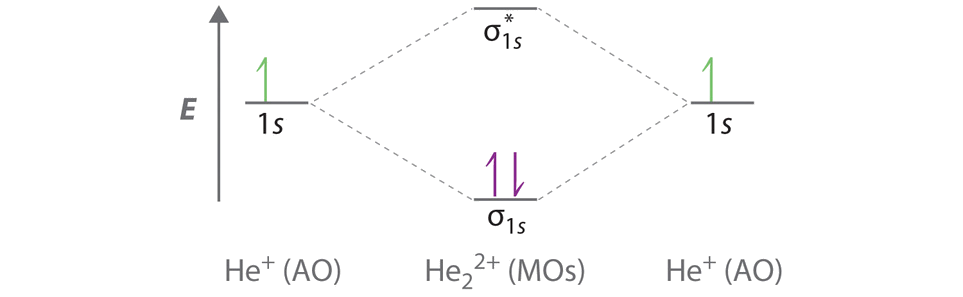
Use the mo diagram provided below to answer the following questions. To get a negative charge on o2 you would have to have a single bond so bond order is 1. Draw the mo for o 2. Barium and chlorine e.
The best way to learn how to draw mo diagrams is to work on practice problems. How can i use molecular orbital diagrams to predict bond order and species diamagnetic or paramagnetic. Use the diagram below to answer the following questions.
Select what is the bond order for n. How can i use molecular orbital diagrams to predict bond order and species diamagnetic or paramagnetic. Draw the mo diagram for.
Select is n2 paramagnetic or diamagnetic select netic. Use this mo diagram to answer the questions below according to molecular orbital theory which of the following species is the most likely to exist ie which will have the greatest bond order. 155 please select the best answer from the choices provided.
Aluminum and sulfur d. There are two mo diagrams you need to memorize for diatoms n2 o2 ne2 etc. Write formulas for compounds formed from the following elements and provide a compound name.
Use the mo diagram provided below to answer the following questions. Silver i and chlorine 7. C2 is unstable diatomic carbon and if it exists it will have a double bond and a bond order of 2 lewis structures are a bit sketchy for these.
The first photo is straight from a 2006 edition pearson general chemistry textbook and it shows you what the molecular orbital mo diagram for o2 is. What is the bond order for n. Then just fill the.
From that diagram you can then easily fill out what the o2 and o2 mo diagrams should beand that is in the second photo i included. Assuming o2 still has a double bond then bond order is 2 bond order is 3 in n2 since it has a triple bond. Use the diagram below to answer the following questions.
Try the following mos on your own and then check with the answers provided. Complete the equation for the reaction of nahco3s with hc2h3o2aq. Sodium and bromine b.
One is for the elements up to nitrogen. Draw the mo diagram for n2. The other is for after nitrogen starting at oxygen.
Potassium and oxygen c. What phase changes occur when the temperature is held constant at 140c and the pressure is increased from 025 atm to 14 atm. However recall that the more electronegative atom will be lower on the diagram.
Lithium and oxygen f. Analyze the diagram below and complete the instructions that follow. Ln bisects klm into two congruent angles measuring 3x 4 and 4x 27 find mklm.
A n2 b n2.
 Molecular Orbital Diagram Wikipedia
Molecular Orbital Diagram Wikipedia
 Using The Mo Diagram Of No Calculate The Bond Order
Using The Mo Diagram Of No Calculate The Bond Order
 Solved 5 In The Space Provided Below Answer The Followin
Solved 5 In The Space Provided Below Answer The Followin
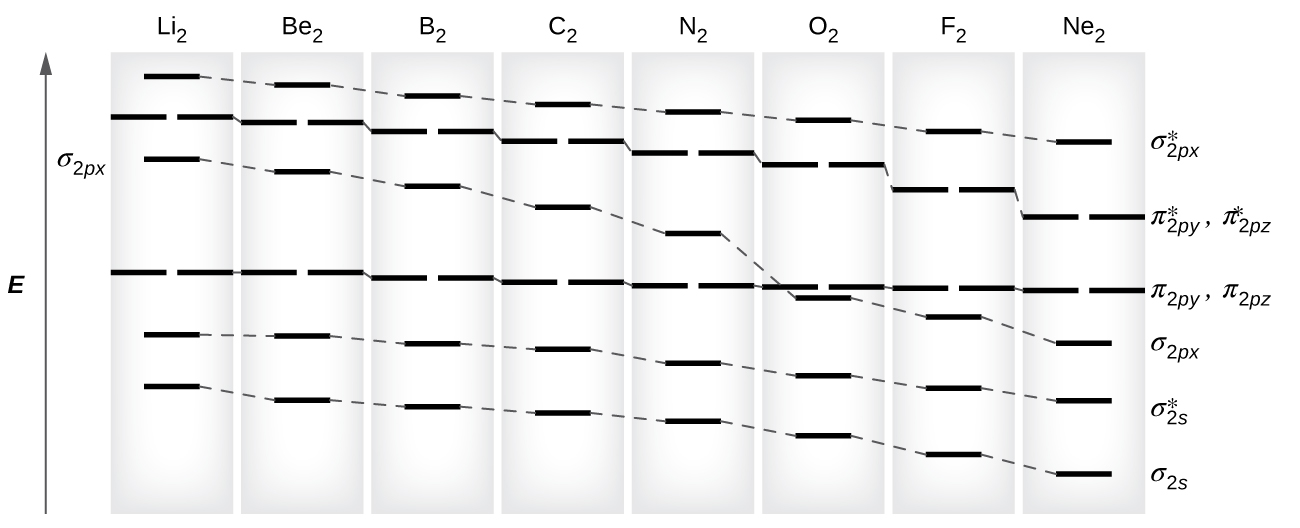 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
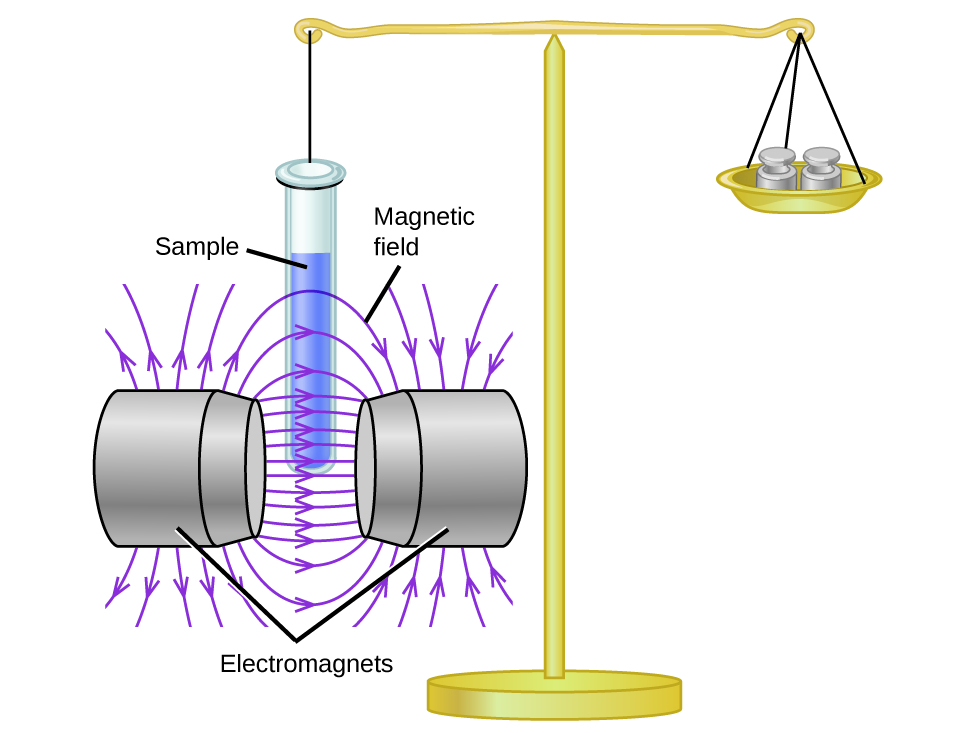 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 Pictorial Molecular Orbital Theory Chemistry Libretexts
Pictorial Molecular Orbital Theory Chemistry Libretexts
 Why Is The Nitrogen S Lone Pair Part Of Sp2 Orbital In
Why Is The Nitrogen S Lone Pair Part Of Sp2 Orbital In
 74 Questions With Answers In Molecular Orbital Theory
74 Questions With Answers In Molecular Orbital Theory
74 Questions With Answers In Molecular Orbital Theory
 Mo Diagram For N2 Molecular Orbital
Mo Diagram For N2 Molecular Orbital
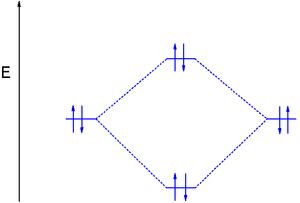 Molecular Orbital Diagram Wikipedia
Molecular Orbital Diagram Wikipedia
 74 Questions With Answers In Molecular Orbital Theory
74 Questions With Answers In Molecular Orbital Theory
Teach Yourself Phase Diagrams And Phase Transformations
What Is The Molecular Orbital Diagram For O2 And O2 Ions
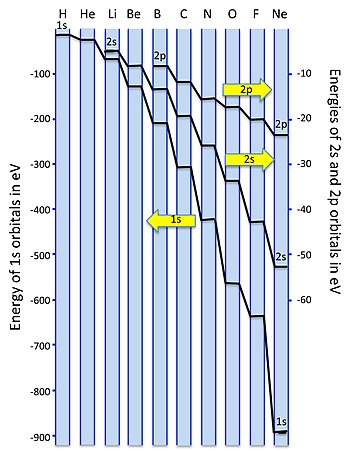 Introduction To Inorganic Chemistry Molecular Orbital Theory
Introduction To Inorganic Chemistry Molecular Orbital Theory
 9 7 Molecular Orbitals Chemistry Libretexts
9 7 Molecular Orbitals Chemistry Libretexts
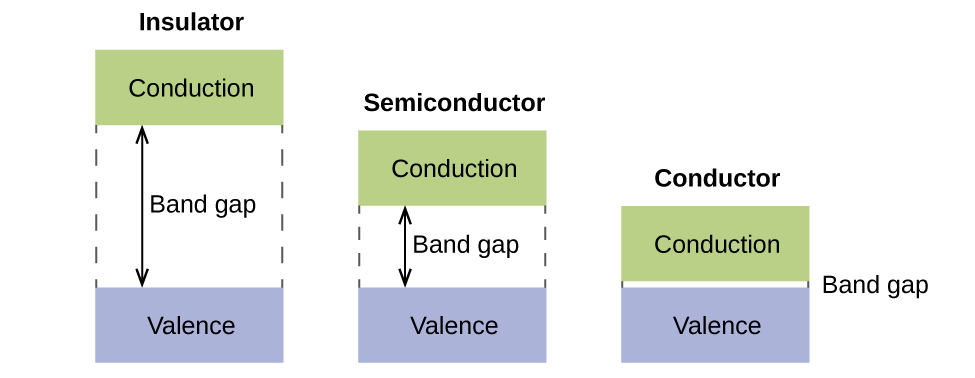 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 Solved 4 In The Space Provided Below Answer The Followin
Solved 4 In The Space Provided Below Answer The Followin
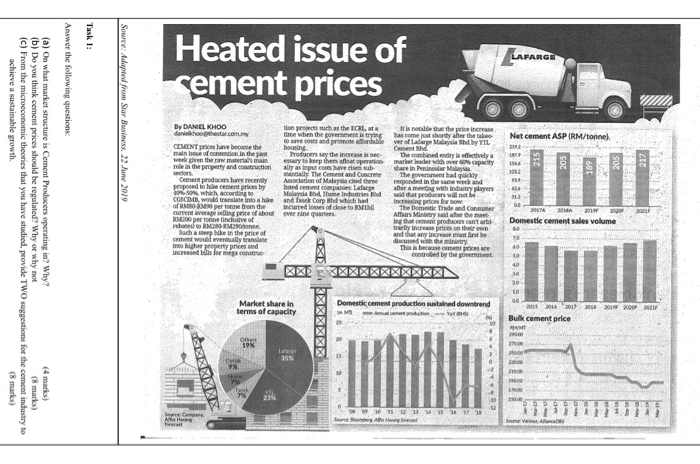 Economics Recent Questions Chegg Com
Economics Recent Questions Chegg Com
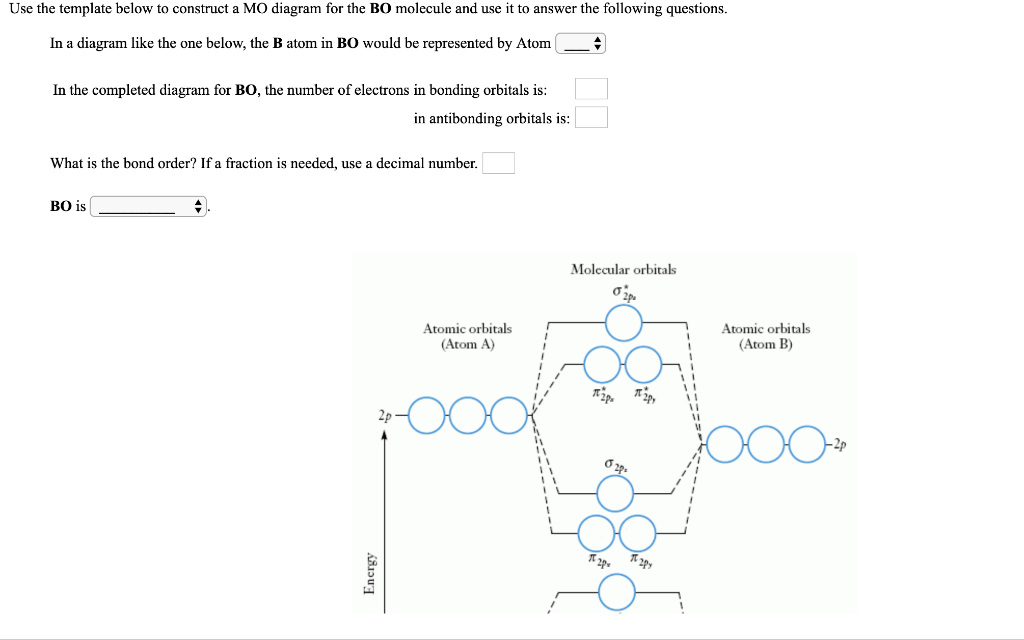 Solved Use The Template Below To Construct A Mo Diagram F
Solved Use The Template Below To Construct A Mo Diagram F
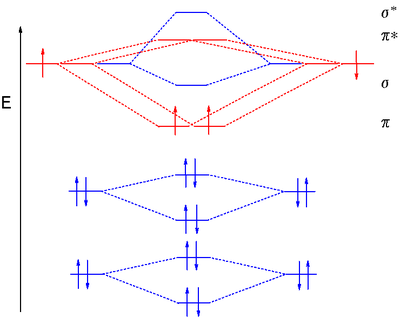 Molecular Orbital Diagram Wikipedia
Molecular Orbital Diagram Wikipedia
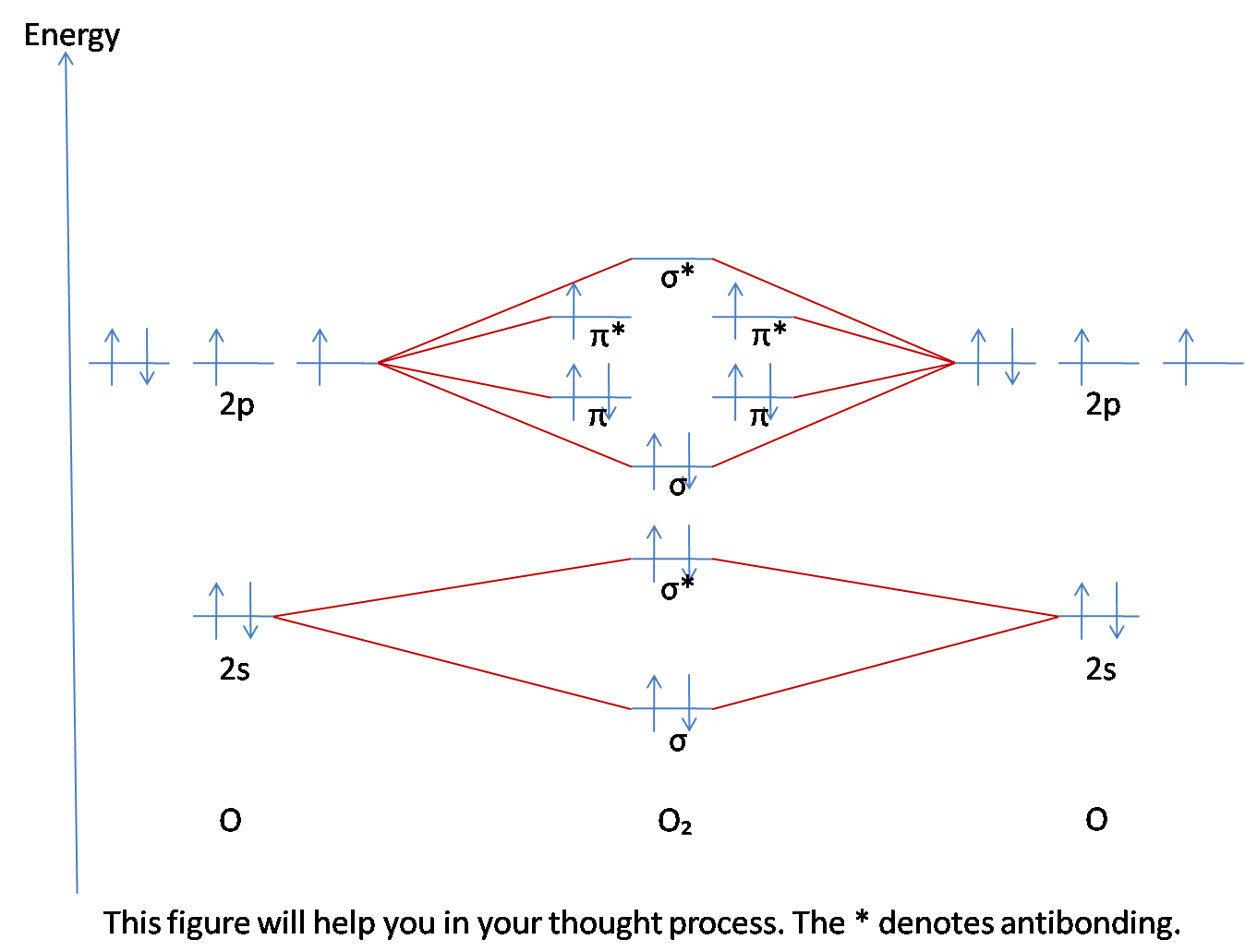 Contrasting Mo And Vb Theory Chemistry Libretexts
Contrasting Mo And Vb Theory Chemistry Libretexts
Belum ada Komentar untuk "Use The Mo Diagram Provided Below To Answer The Following Questions"
Posting Komentar