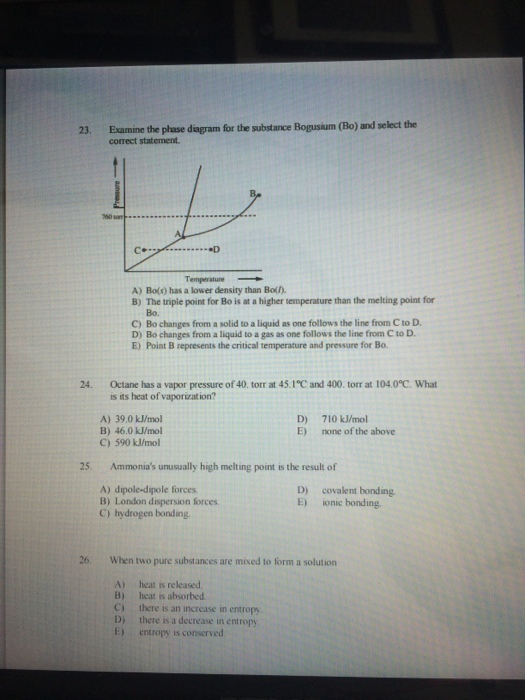
Examine The Phase Diagram For The Substance Bogusium Bo And Select The Correct Statement
Abos has a lower density than bol. Bo changes from a solid to a liquid as one follows the line from c to d.
The triple point for bo is at a higher temperature than the melting point for bo.
Examine the phase diagram for the substance bogusium bo and select the correct statement. A bo has a lower density than bo. Examine the phase diagram for the substance bogusium bo and select the correct statement. Sl chem 101 exam 4.
Examine the phase diagram for the substance bogusium bo and select the correct statement. Examine the phase diagram for the substance bogusium bo and select the correct statement. Examine the phase diagram for the substance bogusium bo and select the correct statement.
Examine the following phase diagram and identify the feature represented by point a. Torr at 104 0 degree c. Bthe triple point for bo is at a higher temperature than the melting point for bo.
9 examine the phase diagram for the substance bogusium bo and select the correct statement. A bos has a lower density than bol. C bo changes from a solid to a liquid as one follows the line from c to d.
C bo changes from a solid to a liquid as one follows the line from c to d. E point b represents the critical temperature and pressure for bo. Examine the phase diagram for the substance bogusium bo and select the correct statement.
A 0145 24 examine the phase diagram for the substance. B the triple point for bo is at a higher temperature than the melting point for bo. B the triple point for bo is at a higher temperature than the melting point for bo.
This preview has intentionally blurred sections. A bos has a lower density than bol. Show transcribed image text 23.
Examine the phase diagram for the substance bogusium bo and select the correct statement. Bo changes from a liquid to a gas as one follows the line from c to d. Sign up to view the full version.
Torr at 451 degree c and 400. Examine the phase diagram for the substance bogusium bo and select the correct statement. Examine the phase diagram for the substance bogusium bo and select the correct statement.
Bo s has a lower density than bo l. Octane has a vapor pressure of 40. On heating the iron and oxygen react according to the balanced reaction below.
Bo changes from a solid to a liquid as one follows the line from c to d. The triple point for bo is at a higher temperature than the melting point for bo. B the triple point for bo is at a higher temperature than the melting point for bo.
A 224 l high pressure reaction vessel is charged with 03910 mol of iron powder and 12 atm of oxygen gas at standard temperature. Point b represents the critical temperature and pressure for bo. Cbo changes from a solid to a liquid as one follows the line from c to d.
Dbo changes from a liquid to a gas as one.
Name Date 1 What Is The Molarity Of A Solution
 Quiz 1 Quiz 1 1 Examine The Following Phase Diagram And
Quiz 1 Quiz 1 1 Examine The Following Phase Diagram And
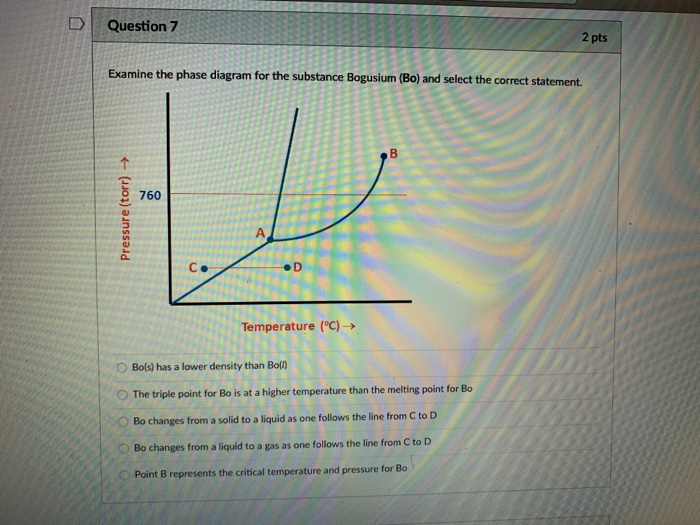
 A 0145 24 Examine The Phase Diagram For The Substance
A 0145 24 Examine The Phase Diagram For The Substance
 A 0145 24 Examine The Phase Diagram For The Substance
A 0145 24 Examine The Phase Diagram For The Substance
 H 2 O Answer Gas Liquid Solid Supercritical Fluid Problem
H 2 O Answer Gas Liquid Solid Supercritical Fluid Problem
 Examine The Following Phase Diagram And Determine What Phase
Examine The Following Phase Diagram And Determine What Phase

 Solved Which Of The Following Should Have The Highest Sur
Solved Which Of The Following Should Have The Highest Sur
 A 0145 24 Examine The Phase Diagram For The Substance
A 0145 24 Examine The Phase Diagram For The Substance
 Quiz 1 Quiz 1 1 Examine The Following Phase Diagram And
Quiz 1 Quiz 1 1 Examine The Following Phase Diagram And
高雄醫學大學 108 學年度學士後醫學系招生考試試題 科目 物理及
 2 Are Nonpolar Draw The Lewis Structure Of The Following
2 Are Nonpolar Draw The Lewis Structure Of The Following
高雄醫學大學 108 學年度學士後醫學系招生考試試題 科目 物理及
 Examine The Phase Diagram For The Substance Bogusium Bo And
Examine The Phase Diagram For The Substance Bogusium Bo And
高雄醫學大學 108 學年度學士後醫學系招生考試試題 科目 物理及
Exam 2017 Chm 132 General Chemistry Ii Studocu



Belum ada Komentar untuk "Examine The Phase Diagram For The Substance Bogusium Bo And Select The Correct Statement"
Posting Komentar