Study The Following Reaction Energy Diagram
Answer to study the following reaction energy diagram. Products energy actan then answer the following questions about the chemical reaction release absorb neither does this reaction release or absorb energy.
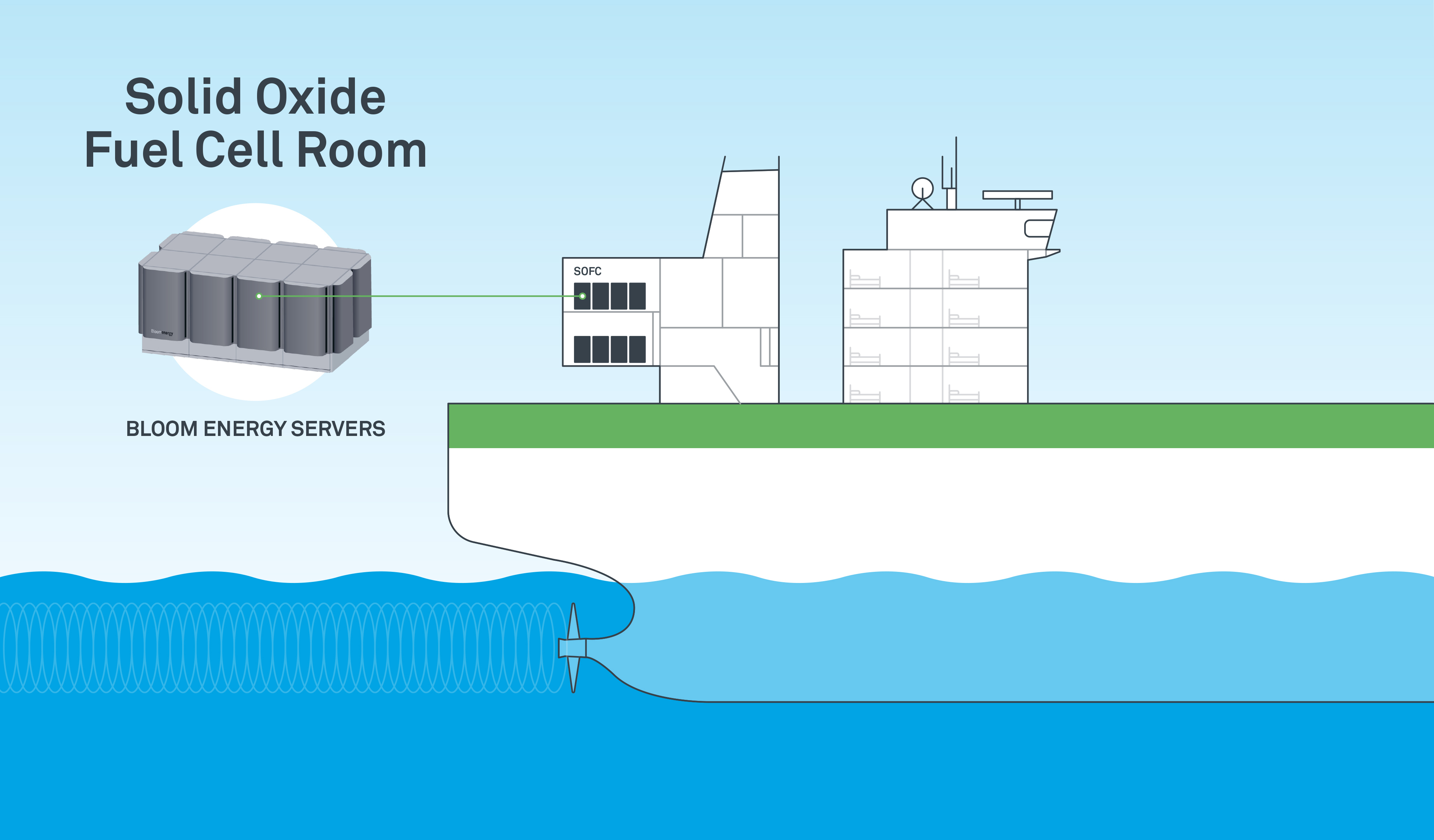 Bloom Energy And Samsung Heavy Industries Team Up To Build
Bloom Energy And Samsung Heavy Industries Team Up To Build
Reaction energy diagram is the graph of reaction progress versus energy.
Study the following reaction energy diagram. Note that the activation energy between reactant and the intermediate step 1 δ g 1 is greater than the activation energy between the intermediate and the products step 2 δ g 2. Ea forward ea reverse if deltah0 which of the following statements correctly describe the key aspects of drawing a reaction energy diagram. Draw the structure of reactants and products on the diagram.
Based on the following energy diagram the heat of reaction for the reaction represented is approximately 20 kcal. The reaction intermediate occurs between two transition states however its energy is still higher than either products or reactants. Which of the following is an energy diagram for a two step reaction which of from chem 22206 at hunter college cuny.
Products energy then answer the following questions about the chemical reac. Label the axes the ea the δ h and the transition state of the reaction. Assume the reaction is exothermic and δ h 75 kjmol and e a 50 kjmol.
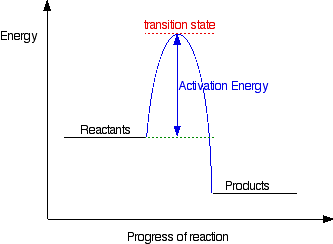
For an exothermic reaction the energy of the products is less than the energy of the reactants. How many transition states occur during yes could this be an elementary reaction. The vertical axis represents the overall energy of the reactants and the horizontal axis represents the reaction progress.
Draw an energy diagram for the following s n 2 reaction. Identify which points on sample reaction coordinate diagrams represent the activation energy and the change in energy discuss what is plotted on the x and y axes of a reaction coordinate diagram. No if you said this reaction could not be elementary then how.
C the stoichiometry of the reaction moles ratio of reactants and products must be known to relate rate. Study the following reaction energy diagram. B rates depend on concentrations of reactants surface area of reactants temperature and presence of catalyst.
The above reaction is a propagation reaction as we see a radicalch3 in the reactant has reacted to form a free radical in the productcl. Rates and mechanisms of chemical reactions. A reaction rate is the change in the amount of products or reactants in a given amount of time.
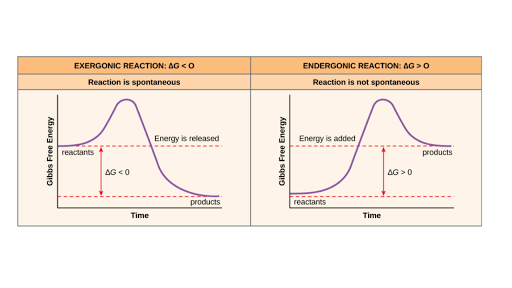
/endothermic-and-exothermic-reactions-602105_final-c4fdc462eb654ed09b542da86fd447e2.png) Endothermic And Exothermic Chemical Reactions
Endothermic And Exothermic Chemical Reactions
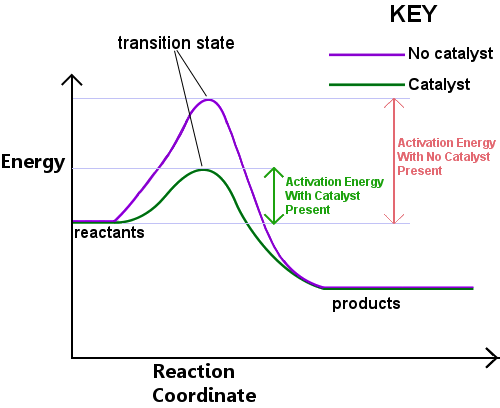 Definition Of Catalyst Chemistry Dictionary
Definition Of Catalyst Chemistry Dictionary
Light Reaction Reaction Steps Photosynthesis With Examples
 A Look At Energy Profiles For Reactions Chemistry Libretexts
A Look At Energy Profiles For Reactions Chemistry Libretexts
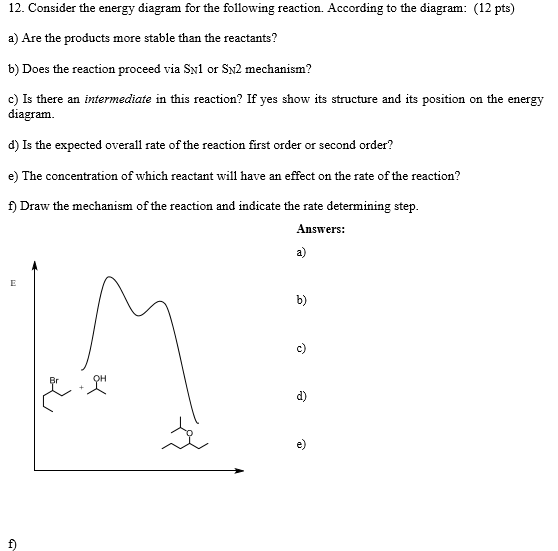 Solved Consider The Energy Diagram For The Following Reac
Solved Consider The Energy Diagram For The Following Reac
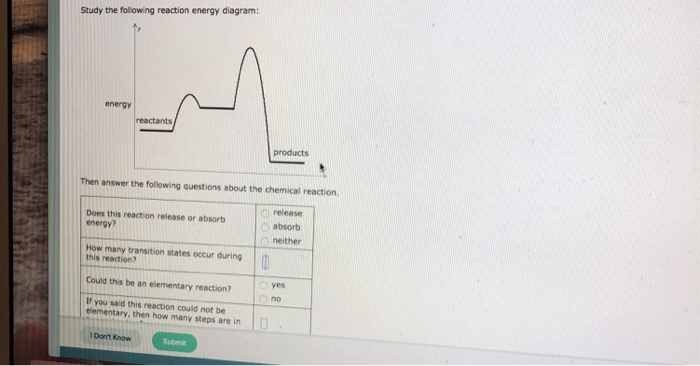 Solved Study The Following Reaction Energy Diagram Energy
Solved Study The Following Reaction Energy Diagram Energy
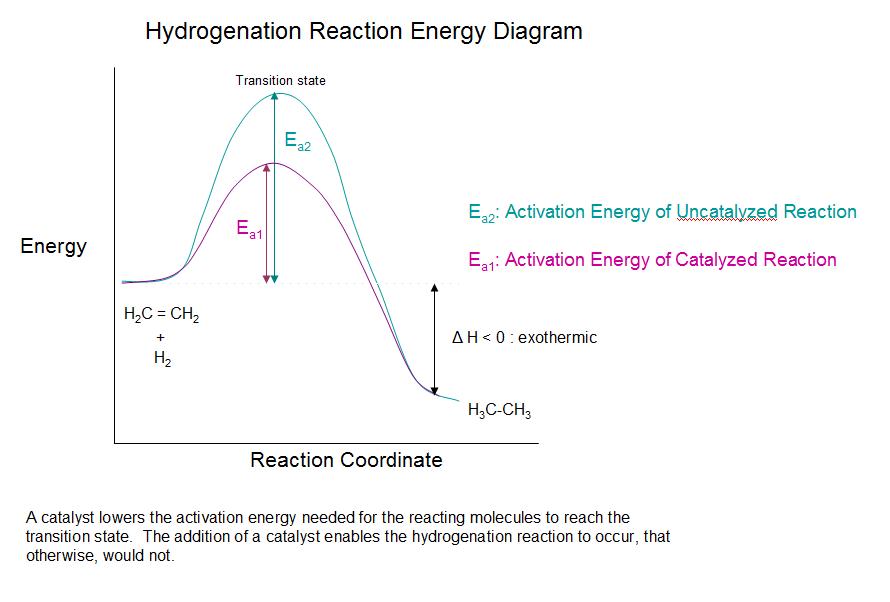 Catalytic Hydrogenation Of Alkenes Chemistry Libretexts
Catalytic Hydrogenation Of Alkenes Chemistry Libretexts
 Quiz Worksheet Energy Reaction Coordinate Diagrams
Quiz Worksheet Energy Reaction Coordinate Diagrams
 The Sn1 Mechanism Energy Diagram Stereochemistry With
The Sn1 Mechanism Energy Diagram Stereochemistry With
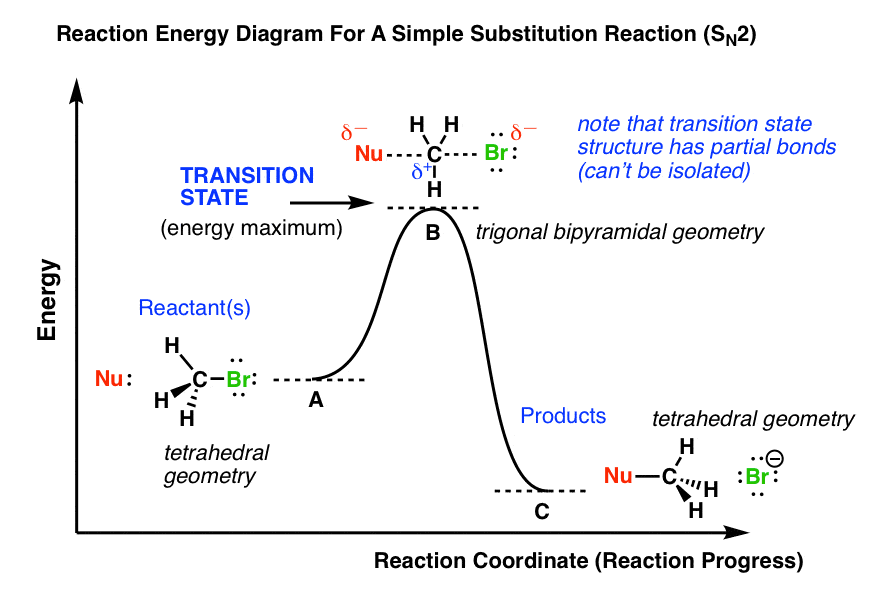
 The Sn2 Mechanism Energy Diagram Mechanism And Stereochemisy
The Sn2 Mechanism Energy Diagram Mechanism And Stereochemisy
 Quiz Worksheet Energy Reaction Coordinate Diagrams
Quiz Worksheet Energy Reaction Coordinate Diagrams
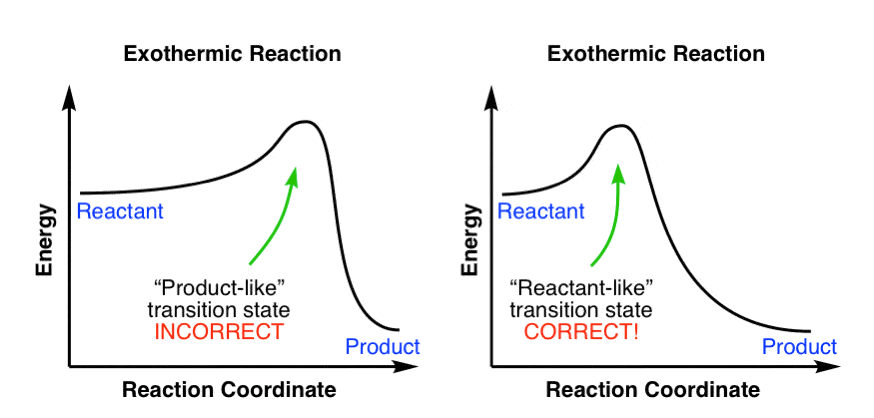
 Hammond S Postulate Master Organic Chemistry
Hammond S Postulate Master Organic Chemistry
:max_bytes(150000):strip_icc()/catalystenergydiagram-56a12b265f9b58b7d0bcb2fe.jpg) Reaction Rate Constant Definition And Equation
Reaction Rate Constant Definition And Equation
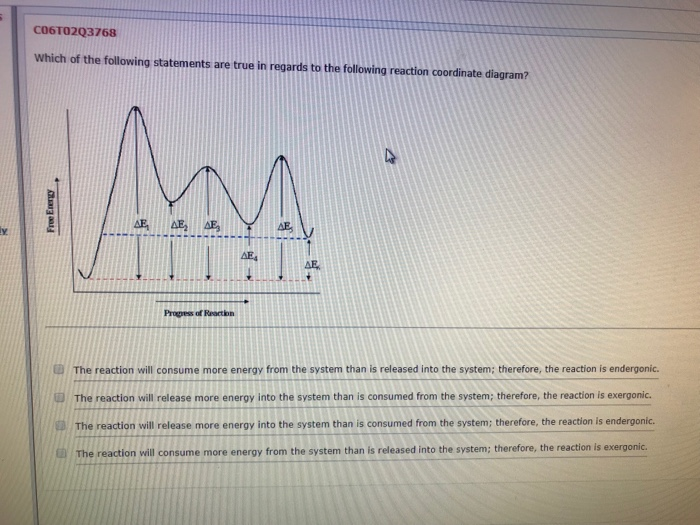 Wileyplus Com Edugen Student Mainfr Un Klein Orga
Wileyplus Com Edugen Student Mainfr Un Klein Orga
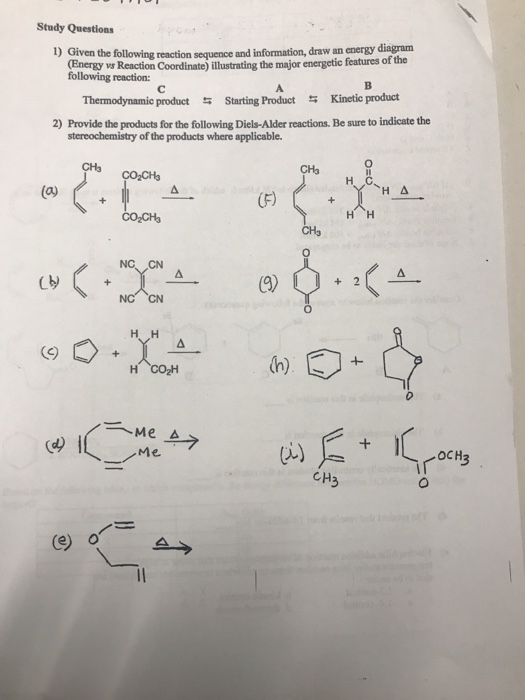
 Solved 3 30 Pts Study The Following Reaction Scheme O
Solved 3 30 Pts Study The Following Reaction Scheme O
 Free Energy An Overview Sciencedirect Topics
Free Energy An Overview Sciencedirect Topics
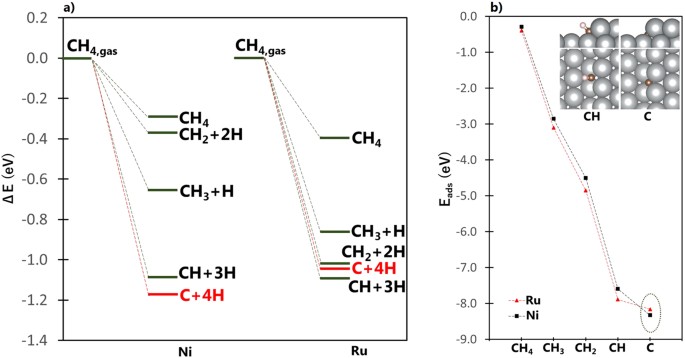 Tuning Methane Decomposition On Stepped Ni Surface The Role
Tuning Methane Decomposition On Stepped Ni Surface The Role
 Free Energy An Overview Sciencedirect Topics
Free Energy An Overview Sciencedirect Topics
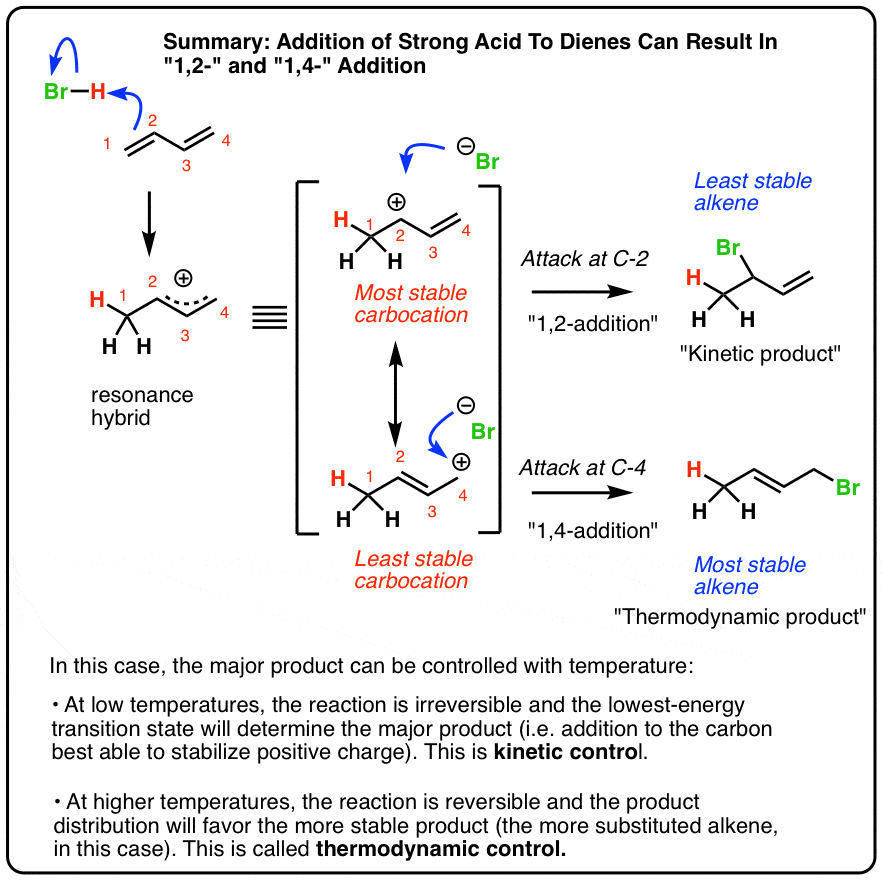 Reactions Of Dienes 1 2 And 1 4 Addition Master Organic
Reactions Of Dienes 1 2 And 1 4 Addition Master Organic
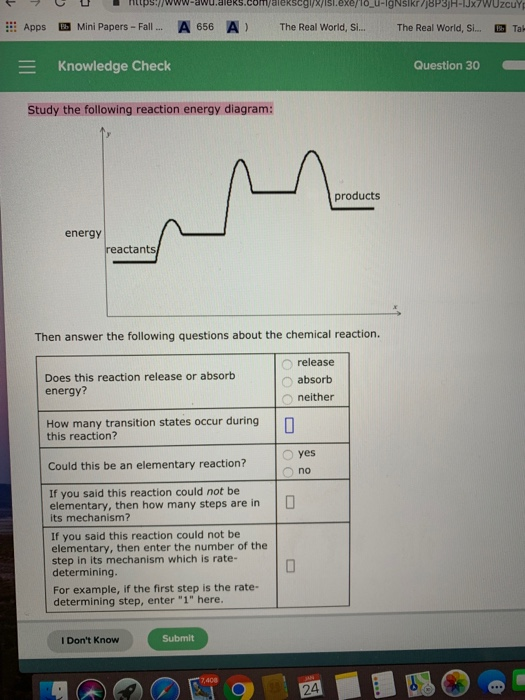 Solved Appsmini Papers Fall A 656 A The Real World S
Solved Appsmini Papers Fall A 656 A The Real World S
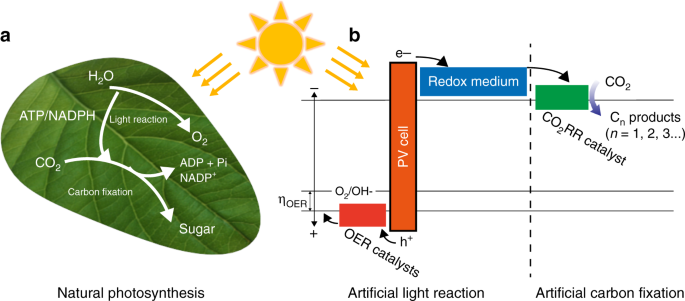 Efficient Solar Driven Electrocatalytic Co 2 Reduction In A
Efficient Solar Driven Electrocatalytic Co 2 Reduction In A
 Chlorination Energy Diagram Wiring Diagram Schematics
Chlorination Energy Diagram Wiring Diagram Schematics


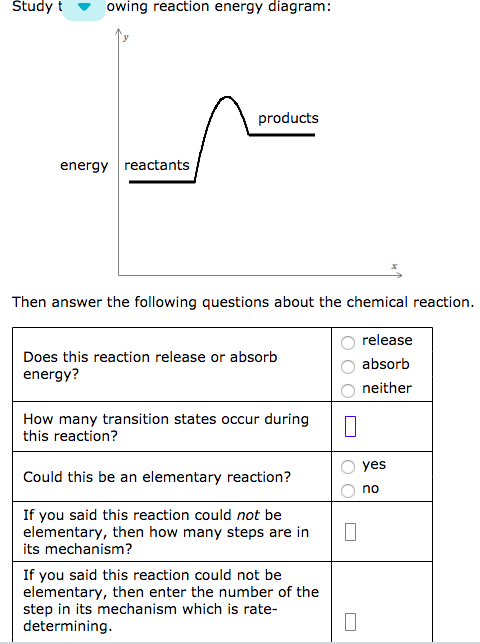 Solved N Energy Diagram Products Energy Reactants Then A
Solved N Energy Diagram Products Energy Reactants Then A

Belum ada Komentar untuk "Study The Following Reaction Energy Diagram"
Posting Komentar