Lewis Dot Diagram For Ch4
The lewis dot structure for nh3 ammonia is shown above. What is the lewis dot structure for ch4.
Dot Diagram Co Catalogue Of Schemas
Methane for the ch4 lewis structure calculate the total number of valence electrons for the ch4 molecule ch4 has 8.

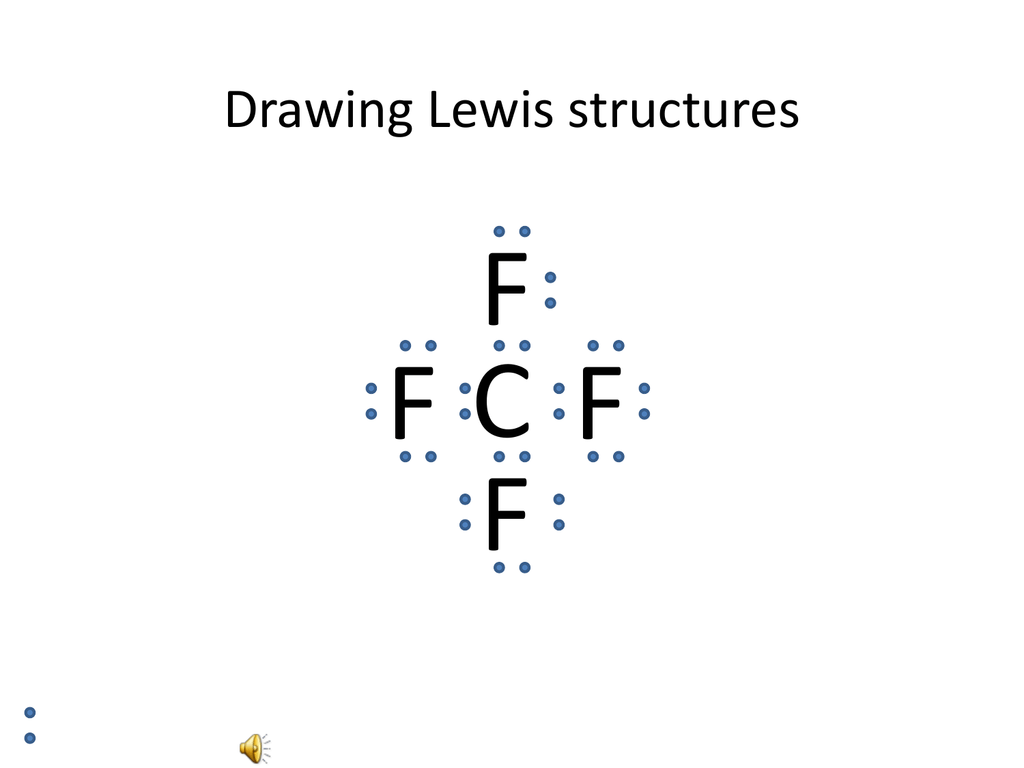
Lewis dot diagram for ch4. The ch 4 lewis structure is one of the most frequently tested lewis structures. Drawing the lewis structure for ch 4 named methane requires only single bondsits one of the easier lewis structures to draw. Remember that hydrogen atoms always go on the outside of a lewis structure and that they only need two valence electrons for a full outer shell.
Drawing the lewis structure for ch 4. The lewis dot structure for ch4 is shown above. In the formal way we find how many electrons we have step 1 how many each atom needs step 2 how many of those are bonding step 3 4 and how many are lone pairs step 5.
The lewis structure for li is li with one dot to the right of the element. The lewis structure or lewis dot diagram shows the bonding between atoms of a molecule and any electrons that may exist. These kinds of structures can also be shown by representing each of the bonds with two dots.
There is an easy way and a formal way to draw the lewis structure of ch 4. I would be lazy and look it up on the internet. Lewis structures help us to determine how atoms within a molecule are connected ie.
Note that hydrogen atoms always go on the outside of a lewis dot structure. But seriously you have an electron pair between the c and each of the hs in the lewis diagram a ala why is that the correct diagram you ask. For ch 4 you have a total of 8 total valence electrons.
A step by step explanation of how to draw the ch4 lewis dot structure. This info can then be used to determine the lewis dot structure. Explains how to draw the lewis dot structure for ch 4 methane.
Which atom connects to which and using how many bonds. This is because they can share a maximum of two electrons. Each atom in the bond has a full valence with carbon having access to eight electrons and each hydrogen having access to two this is why hydrogen only needs two.
First each hydrogen has only one electron to donate or. You could also represent the bonds as dots between the two atoms but this may be confused with the lone pair electrons on the nitrogen.
 Lewis Dot Structure Practice Flashcards Quizlet
Lewis Dot Structure Practice Flashcards Quizlet
 Draw The Lewis Structure For Methane Ch4 Ppt Video
Draw The Lewis Structure For Methane Ch4 Ppt Video
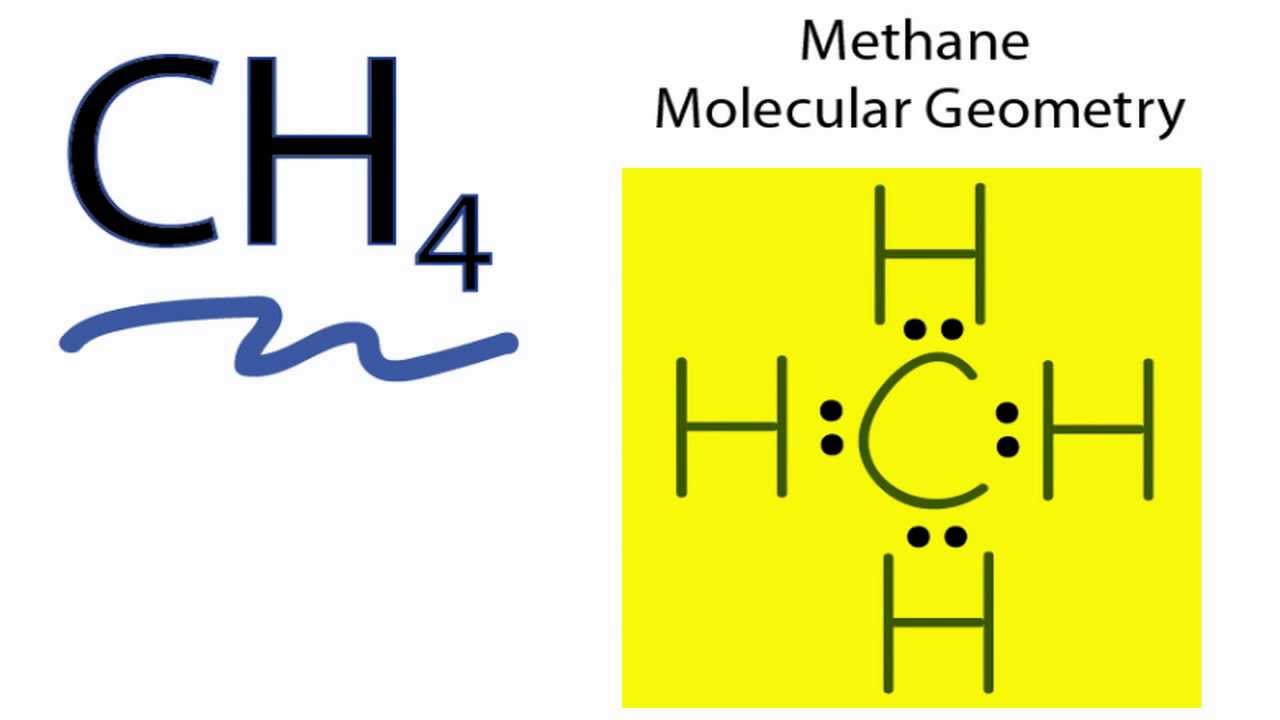 Ch4 Molecular Geometry Shape And Bond Angles
Ch4 Molecular Geometry Shape And Bond Angles
 Explain The Formation Of Ch4 Molecule With Lewis Dot
Explain The Formation Of Ch4 Molecule With Lewis Dot
 The Lewis Dot Structure For Ch4 Makethebrainhappy
The Lewis Dot Structure For Ch4 Makethebrainhappy
 Introduction Covalent Bonds Form Between Nonmetals The Number
Introduction Covalent Bonds Form Between Nonmetals The Number
 How To Draw Methane Ch4 Lewis Structure
How To Draw Methane Ch4 Lewis Structure
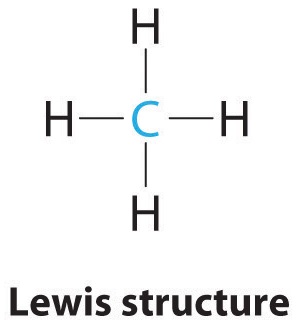 9 7 The Shapes Of Molecules Chemistry Libretexts
9 7 The Shapes Of Molecules Chemistry Libretexts
 Lewis Structures Single Double Triple Bonds Video
Lewis Structures Single Double Triple Bonds Video
Topic Lewis Dot Diagrams For Ionic Compounds
 Quiz Worksheet Lewis Structures Study Com
Quiz Worksheet Lewis Structures Study Com
 Ch4 Lewis And 3 D Structures Dr Sundin Uw Platteville
Ch4 Lewis And 3 D Structures Dr Sundin Uw Platteville
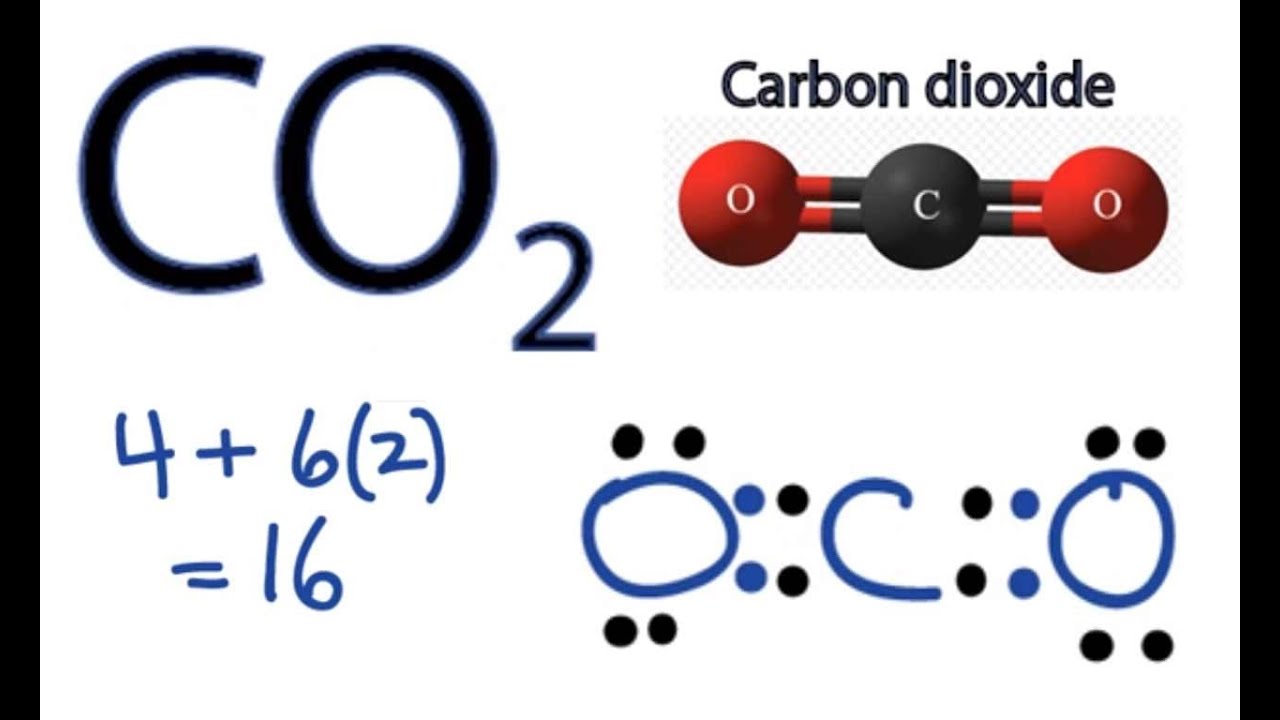 Co2 Lewis Structure How To Draw The Dot Structure For Carbon Dioxide
Co2 Lewis Structure How To Draw The Dot Structure For Carbon Dioxide
 Ch4 Lewis Structure How To Draw The Dot Structure For Ch4
Ch4 Lewis Structure How To Draw The Dot Structure For Ch4
 How Can The Electron Dot Structure For Ch4 Be Determined
How Can The Electron Dot Structure For Ch4 Be Determined
 Solved Draw The Lewis Structure Of Methane Ch4 Which Is
Solved Draw The Lewis Structure Of Methane Ch4 Which Is
 What Is The Vsepr Shape Of The Molecule Ch4 Socratic
What Is The Vsepr Shape Of The Molecule Ch4 Socratic
 How Can The Electron Dot Structure For Ch4 Be Determined
How Can The Electron Dot Structure For Ch4 Be Determined
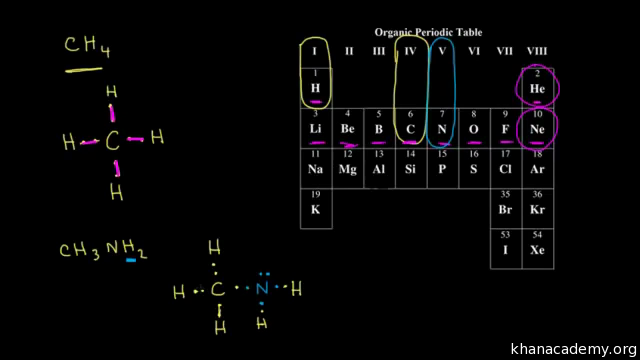 Dot Structures I Single Bonds Video Khan Academy
Dot Structures I Single Bonds Video Khan Academy
 Wedge And Dash Projection Definition And Example
Wedge And Dash Projection Definition And Example
 The Lewis Dot Structure For Nh3 Makethebrainhappy
The Lewis Dot Structure For Nh3 Makethebrainhappy
 C2h4 Lewis Dot Structure How To Draw The Lewis Structure For C2h4
C2h4 Lewis Dot Structure How To Draw The Lewis Structure For C2h4
 Propyn Wikiwand C2h2 Dot Diagram Ch4 Propan 1 Ol Structure
Propyn Wikiwand C2h2 Dot Diagram Ch4 Propan 1 Ol Structure
 Draw Electron Dot Structure Of Ch4 Brainly In
Draw Electron Dot Structure Of Ch4 Brainly In
 Lewis Dot Structure The Basics Of Chemical Bonding
Lewis Dot Structure The Basics Of Chemical Bonding

Belum ada Komentar untuk "Lewis Dot Diagram For Ch4"
Posting Komentar